Listeria monocytogenes
Listeria is a bacterium that is commonly found in the environment, on our hands and in our refrigerators. Most people are routinely exposed to Listeria with no health consequences. However, one type of Listeria – Listeria monocytogenes – is extremely virulent. L. monocytogenes infections can lead to the very serious disease, listeriosis, particularly among at-risk populations, including pregnant women, newborns, the elderly and those who are immunocompromised.
L. monocytogenes can be found in unprocessed foods of animal origin like raw milk, meat, poultry and fish. L. monocytogenes also can be found in some processed foods like cheese, ice cream and processed meats due to post-processing contamination. These bacteria also are sometimes found on fresh fruits and vegetables. While pasteurization and cooking will destroy L. monocytogenes, on rare occasions this environmental bacterium can recontaminate the food as it is being packaged, handled or distributed.
Listeria can thrive in a cold, moist environment commonly found in refrigerators. Even if a Listeria-free product is placed in a refrigerator, if it is improperly handled or stored, bacteria in the refrigerator can contaminate products. In fact, protein products like meat and cheese are excellent media or “food” for bacterial growth.
Listeriosis
The incubation period for L. monocytogenes ranges from four days to several weeks, which can lead to listeriosis. Listeriosis is a human illness characterized by flu-like symptoms including fever, muscle aches and, sometimes, gastrointestinal symptoms. If infection spreads to the nervous system, symptoms may progress to include severe headache, stiff neck, confusion, loss of balance or convulsions. Once contracted, the symptoms of listeriosis can last for several days. Diagnosis requires laboratory analysis of blood or cerebrospinal fluid. Treatment is administered through antibiotics like penicillin or ampicillin.
Healthy adults and children can become infected, but rarely become seriously ill. Pregnant women may experience only mild flu-like symptoms, but infection during pregnancy can transfer to newborns or lead to premature delivery, miscarriage or stillbirth. According to the Centers for Disease Control and Prevention (CDC), pregnant women are 20 times more likely to contract listeriosis and account for about one-third of reported cases. In such instances, usually the unborn child suffers the most serious effects.
Since 2000, the L. monocytogenes prevalence rate in ready-to-eat meat and poultry products has dropped 80 percent to less than one third of one percent. It is also noteworthy that there have been no recalls of ready-to-eat meat or poultry products triggered by a listeriosis outbreak since 2002.
It is unknown how many of the L. monocytogenes organisms (known as the “infective dose”) are required to cause listeriosis, but it is believed to vary with the specific strain of the bacterium and the susceptibility of the individuals. Research is ongoing on infective dose, but the scientific opinion is currently that low levels of L. monocytogenes are not likely to cause illness in most people.
Incidence of Illnesses
Listeriosis is very rare. Of the estimated 48 million foodborne illnesses that CDC estimates occur each year, listeriosis accounts for a tiny fraction of illnesses (0.017 percent), 2.6 percent of foodborne illness-related hospitalizations and 19 percent of foodborne disease-related deaths. L. monocytogenes decreased from 0.34 cases per 100,000 people in 2009 to 0.28 cases per 100,000 people in 2011.
Recent technological developments have increased the ability of scientists to identify the cause of foodborne illness outbreaks. One in particular being used by CDC, Pulsed Field Gel Electrophoresis (PFGE), analyzes the DNA fingerprints of suspected pathogens from an outbreak to link cases with potential sources of the illness. This provides government agencies and the industry a means by which to identify the source of contamination, reduce the scope of an outbreak and hopefully prevent future outbreaks.
A public health network known as PulseNet allows federal and state government agencies to share data concerning foodborne illness outbreaks, thus improving pathogen tracking and identification capabilities. These developments have and will result in increased awareness of foodborne illness, including listeriosis, within the food industry, the medical and public health communities and the public.
Preventing Listeriosis
The meat and poultry industry controls L. monocytogenes in fully cooked meat and poultry products by achieving a validated lethality step (e.g. cooking). This lethality step is combined with the meat and poultry company having an aggressive environmental control and sanitation program that prevents L. monocytogenes growth in equipment and the environment within the cooked products area of the plant. This control program is one part of a comprehensive food safety process management system that is continually verified. These concepts are the core meat and poultry industry’s preventive food safety system for L. monocytogenes.
Both industry and government are working diligently to reduce the incidence of L. monocytogenes in foods.
Regulatory Oversight
Both the Food Safety and Inspection Service (FSIS) and the FDA have strict regulatory standards for ready-to-eat (fully cooked) products. These standards include a “zero tolerance” for L. monocytogenes on all ready-to-eat foods, the strictest standard in the world.
FSIS and FDA have been sampling ready-to-eat foods in processing plants for L. monocytogenes since 1987. Foods found to contain the organism are either withheld from distribution into the food supply or recalled.
FSIS Regulatory Testing for Listeria monocytogenes in RTE Meat and Poultry Products
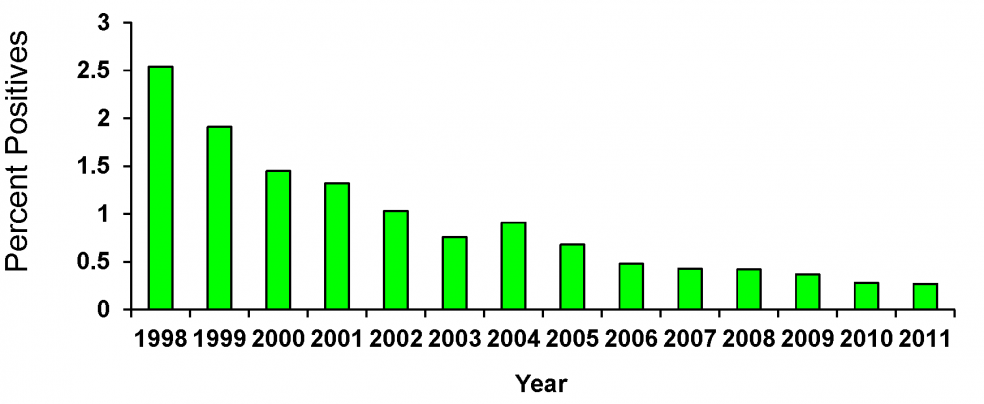
FSIS results of ready-to-eat products analyzed for Listeria monocytogenes – summary by all projects
In recent years, FSIS and industry stepped up their sampling programs for Listeria in processing environments and on ready-to-eat meat and poultry products. FSIS has established regulations that require manufacturers to have environmental sampling and testing programs and have further established risk-based processing alternatives to further eliminate or prevent growth of inadvertent Listeria contamination. The goal of the effort is to monitor the adequacy of the food safety process management system to produce product that does not contain L. monocytogenes.
Consumer Information
The decline in the incidence of L. monocytogenes on ready-to-eat meat and poultry products by FSIS coincides with the transition to a more science-based meat and poultry inspection system in 1998. Despite the meat industry and FSIS efforts in employing safeguards and improvements in processing technologies, post-processing contamination, even in the consumer’s home there is a potential risk for L. monocytogenes contamination. The prevalence of L. monocytogenes in the consumer environment makes proper food handling procedures and sanitation essential for protecting at-risk consumers from this pathogen.
L. monocytogenes, like other bacteria, is very susceptible to heat. Cooking product to 160F for a few seconds is sufficient to kill these bacteria. That is why the meat and poultry industry advises at-risk consumers, such as pregnant women, to follow the CDC’s recommendations: thoroughly reheat to steaming hot ready-to-eat meat and poultry products, like hot dogs and lunchmeats, before consuming them.
Consumers with questions should visit www.meatsafety.org or call USDA’s Meat and Poultry Hotline, 1-888-674-6854.
Helpful Links
- American Meat Institute
- Meat Safety
- American Meat Institute Foundation
- American Society for Microbiology
- Centers for Disease Control and Prevention
- Institute of Food Technologists
Third-Party Experts
Michael Doyle, Ph.D.
Director, Center for Food Safety
University of Georgia
(770) 228-7284
mdoyle@uga.edu
Martin Wiedmann, Ph.D.
Assistant Professor, Department of Food Science
Cornell University
(607) 254-2838
mw16@cornell.edu
